CSA Z463 Electrical Maintenance -
Our customized live online or in‑person group training can be delivered to your staff at your location.

- Live Online
- 6 hours Instructor-led
- Group Training Available
The medical device company said in a letter that 15 patients have received inappropriate electrical jolts from either Cognis or Teligen defibrillators, devices used to treat irregular heartbeats.
As many as 8,000 patients could be at risk, the company said. No deaths have been reported with the issue.
The shocks are mainly due to faulty leads, or wires connecting the devices to the heart, according to the March 23 letter. Not all wires used with the devices are made by Boston Scientific Corp.
Doctors can prevent the problem by turning off a sensor on the devices that can make them overly sensitive to unusual heart signals, states the letter, which was posted to a cardiology Web site.
The sensor records patient's breathing rhythms to give physicians extra information about their condition, though it is not necessary for medical treatment.
"Turning the respiratory sensor off eliminates the possibility of this inappropriate therapy occurring," said a company spokesman in a statement.
Implanted in the upper chest, defibrillators monitor the heart for deadly irregular heartbeats and use electrical jolts to shock it back to normal. They differ from pacemakers, which use lower-voltage electrical pulses to correct dangerously slow heart rhythms.
The approval of the Cognis and Teligen devices last year marked an important milestone for Boston Scientific as the first defibrillators launched under its own name.
The Natick, Mass.-based company entered the heart-regulating business in 2006 with the purchase of Guidant. But the acquisition was marred by controversy surrounding Guidant's Prizm defibrillators, which eventually sank sales for the entire sector. The Indianapolis-based Lilly fixed a flaw with its devices in 2002 but did not inform patients and physicians until three years later.
Citigroup analyst Matthew Dodds said the announcement could pressure product sales in the near term, "but the problem appears easily resolvable and it should not delay further sales."
Related News
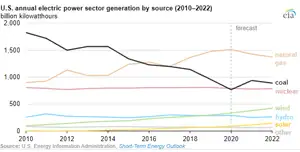
Annual U.S. coal-fired electricity generation will increase for the first time since 2014

Europe's Renewables Are Crowding Out Gas as Coal Phase-Out Slows

Texas produces and consumes the most electricity in the US

Heathrow Airport Power Outage: Vulnerabilities Flagged Days Before Disruption

Frustration Mounts as Houston's Power Outage Extends

PG&E's bankruptcy plan wins support from wildfire victims
Sign Up for Electricity Forum’s Newsletter
Stay informed with our FREE Newsletter — get the latest news, breakthrough technologies, and expert insights, delivered straight to your inbox.
Electricity Today T&D Magazine Subscribe for FREE

- Timely insights from industry experts
- Practical solutions T&D engineers
- Free access to every issue
